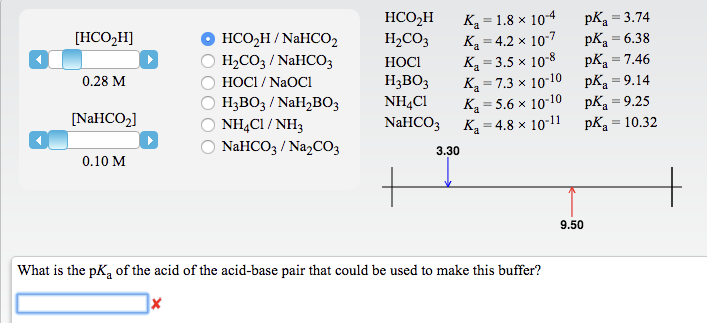
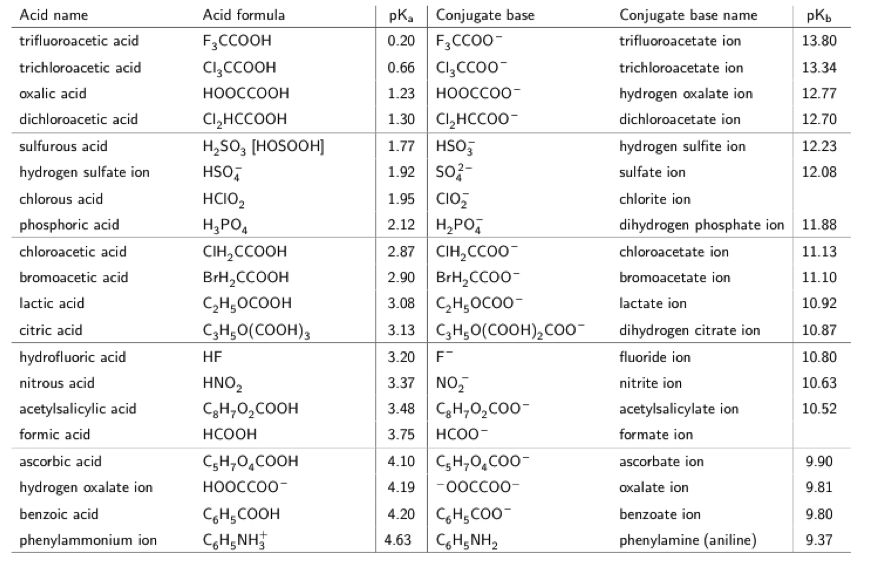
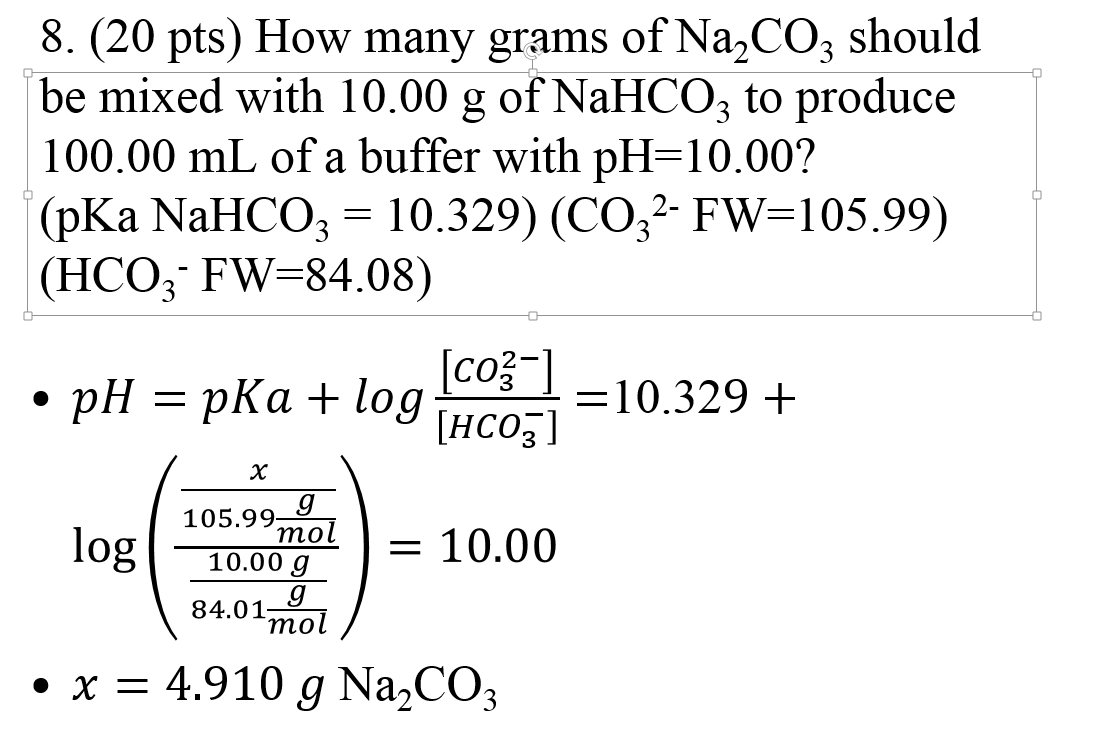The pKa of a weak acid, HA, is 4.80. The pKb of a weak base, BOH, is 4.78. What is the pH of an aqueous solution of the corresponding slat BA?
![SOLVED: A buffer made from NaHCO3 and Na2CO3 is prepared with a pH of 9.40. a. What must the [CO3 ] /[HCO3 ] ratio be? Ka for HCO3 is 4.7 x 10 . SOLVED: A buffer made from NaHCO3 and Na2CO3 is prepared with a pH of 9.40. a. What must the [CO3 ] /[HCO3 ] ratio be? Ka for HCO3 is 4.7 x 10 .](https://cdn.numerade.com/previews/bd62afea-5060-4429-b9f4-4515705d9462_large.jpg)
SOLVED: A buffer made from NaHCO3 and Na2CO3 is prepared with a pH of 9.40. a. What must the [CO3 ] /[HCO3 ] ratio be? Ka for HCO3 is 4.7 x 10 .
Boehm Titration Revisited (Part I): Practical Aspects for Achieving a High Precision in Quantifying Oxygen-Containing Surface Gr
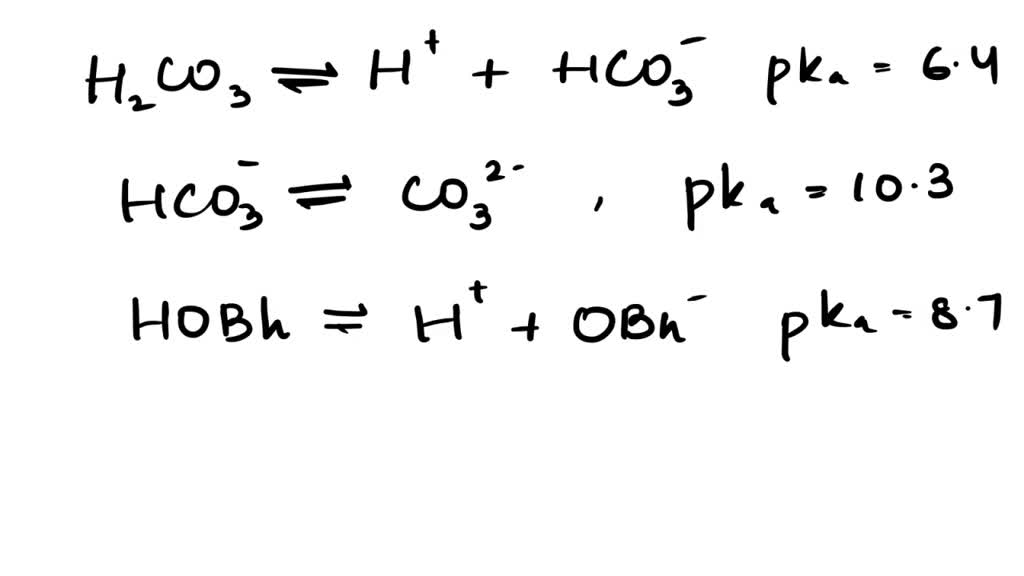
SOLVED: The pKas of H2CO3 are 6.4 10.3. The pKa of HOBr is 8.7. If equimolar amounts of Na2CO3 and HOBr are dissolved in water what will be the predominant anionic species
![50 mL of 0.05 M Na2CO3 is titrated against 0.1 M HCl . On adding 40 mL of HCl , pH of the solution will be[Given for H2CO3, pKa1 = 6.35; pKa2 = 10.33; log 3 = 0.477 , log 2 = 0.30 ] 50 mL of 0.05 M Na2CO3 is titrated against 0.1 M HCl . On adding 40 mL of HCl , pH of the solution will be[Given for H2CO3, pKa1 = 6.35; pKa2 = 10.33; log 3 = 0.477 , log 2 = 0.30 ]](https://dwes9vv9u0550.cloudfront.net/images/1766662/06af4a5d-9ecd-450c-9b4d-b90fc83566c2.jpg)
![The pKa values for various precipitants [17]. | Download Scientific Diagram The pKa values for various precipitants [17]. | Download Scientific Diagram](https://www.researchgate.net/publication/339359335/figure/tbl1/AS:860297669640196@1582122356901/The-pKa-values-for-various-precipitants-17.png)