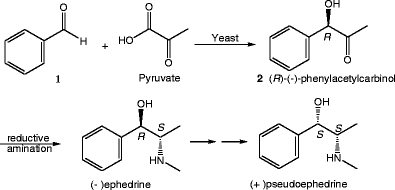
Potential of some yeast strains in the stereoselective synthesis of (R)-(−)- phenylacetylcarbinol and (S)-(+)-phenylacetylcarbinol and their reduced 1,2-dialcohol derivatives | SpringerLink
Potential of some yeast strains in the stereoselective synthesis of (R)-(−)- phenylacetylcarbinol and (S)-(+)-phenylacetylcarbinol and their reduced 1,2-dialcohol derivatives | SpringerLink

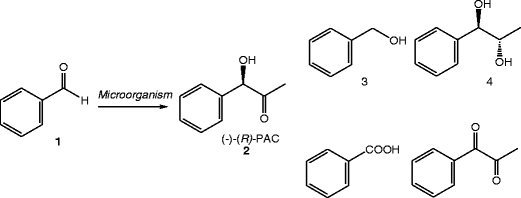
Investigation of the l-phenylacetylcarbinol process to substituted benzaldehydes of interest - ScienceDirect

Figure 1 from Screening of Yeasts for Cell-Free Production of (R)- Phenylacetylcarbinol in a Shake Flask Condition | Semantic Scholar

Production of Ethanol and R-phenylacetylcarbinol from Whole Cell Biocatalyst Utilizes Carbon Source from Dried Longan Amalia S. Agustina, Poonsiri Phrathong, - ppt download
Sciencemadness Discussion Board - Synthesis of Phenylacetylcarbinol by Alkyne Hydration and Subsequent Enamine formation - Powered by XMB 1.9.11

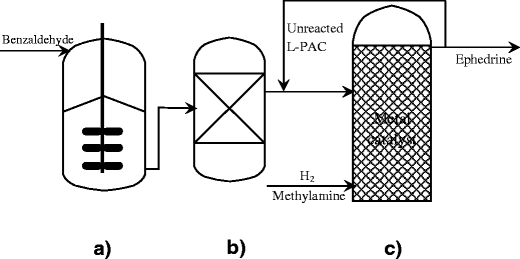
Biotransformation of benzaldehyde to L‐phenylacetylcarbinol (L‐PAC) by Torulaspora delbrueckii and conversion to ephedrine by microwave radiation - Shukla - 2002 - Journal of Chemical Technology & Biotechnology - Wiley Online Library
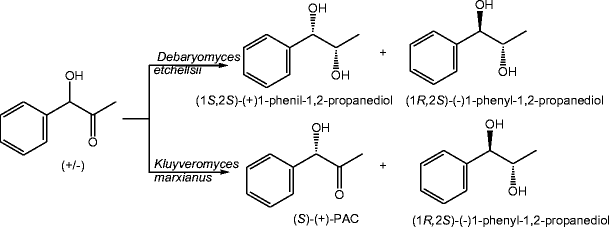
Potential of some yeast strains in the stereoselective synthesis of (R)-(−)- phenylacetylcarbinol and (S)-(+)-phenylacetylcarbinol and their reduced 1,2-dialcohol derivatives | SpringerLink
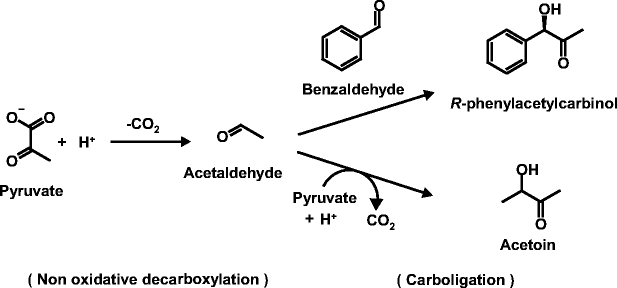
Enzymatic (R)-phenylacetylcarbinol production in a benzaldehyde emulsion system with Candida utilis cells | SpringerLink