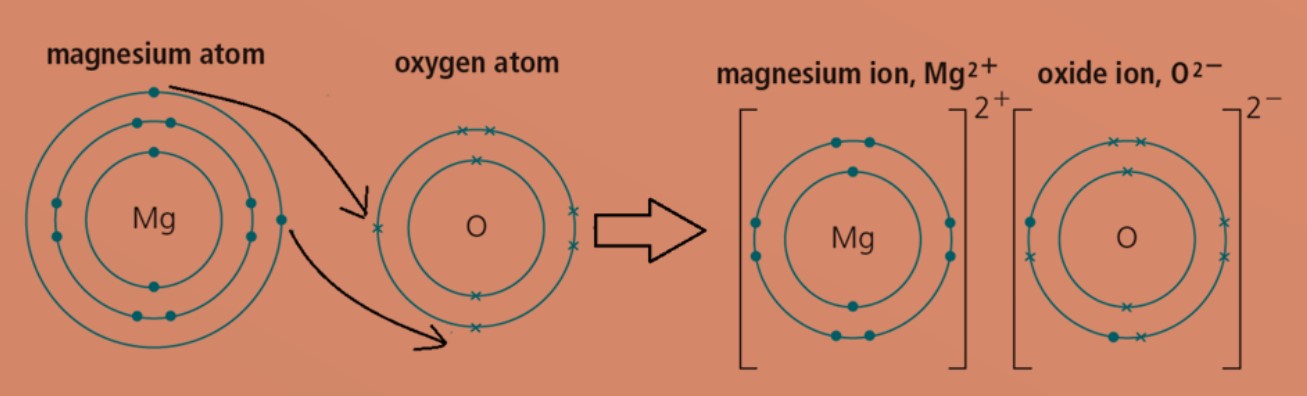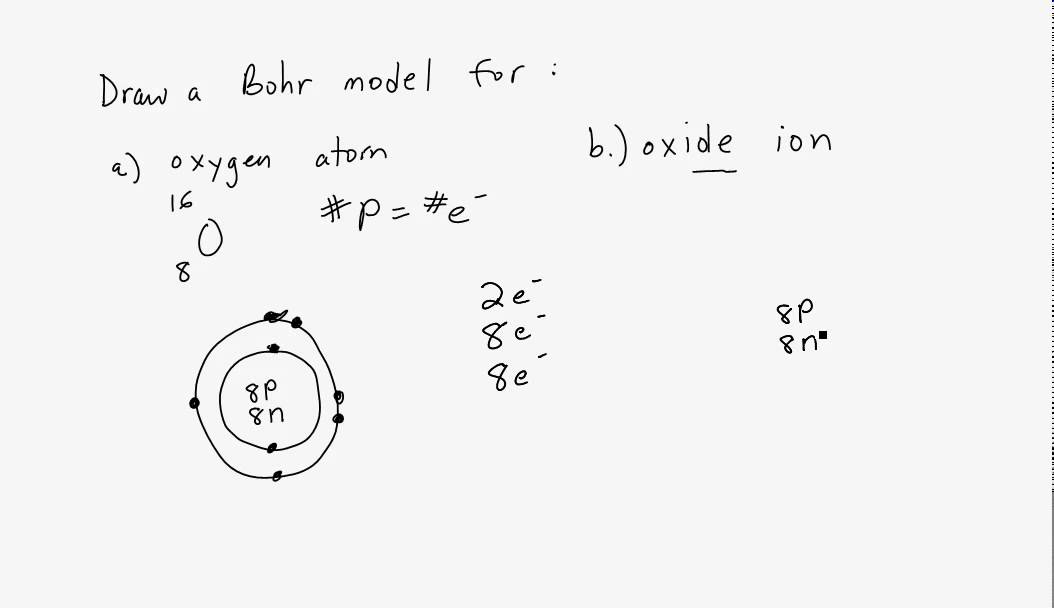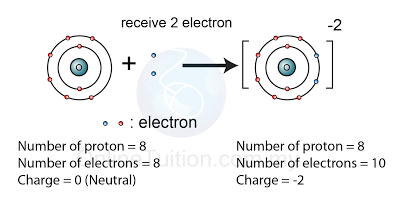
What is the difference between atoms and ions; and covalent compounds and ionic compounds? | Socratic

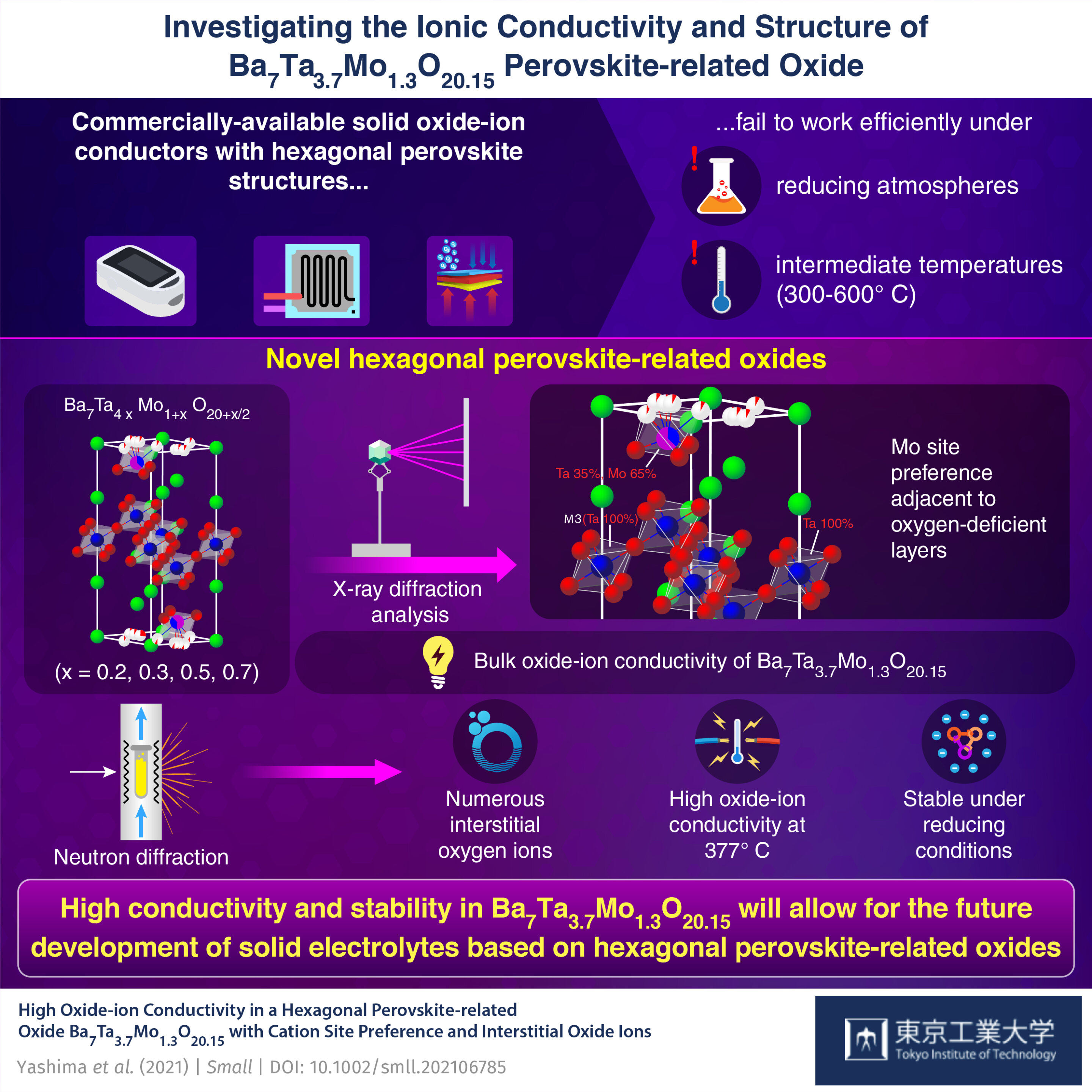
Associating High Oxide-Ion Conductivity and Conduction Mechanisms with Local Atomic Environments in Na0.5Bi0.5–xTi1–yMgyO3−δ | The Journal of Physical Chemistry C

Li2CO3 decomposition in Li-ion batteries induced by the electrochemical oxidation of the electrolyte and of electrolyte impurities - ScienceDirect

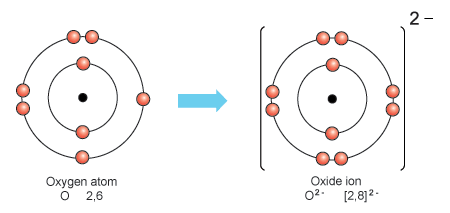
The formation of the oxide ion, O ^2 - (g) , from oxygen atom requires first an exothermic and then an endothermic step as shown below: O(g) + e^-→ O^-(g); Δ H^∘ = -

For the phosphorus (V) tetra oxide ion, draw the Lewis structures for all the most probable resonance structures and assign formal charges next to all atoms. | Homework.Study.com