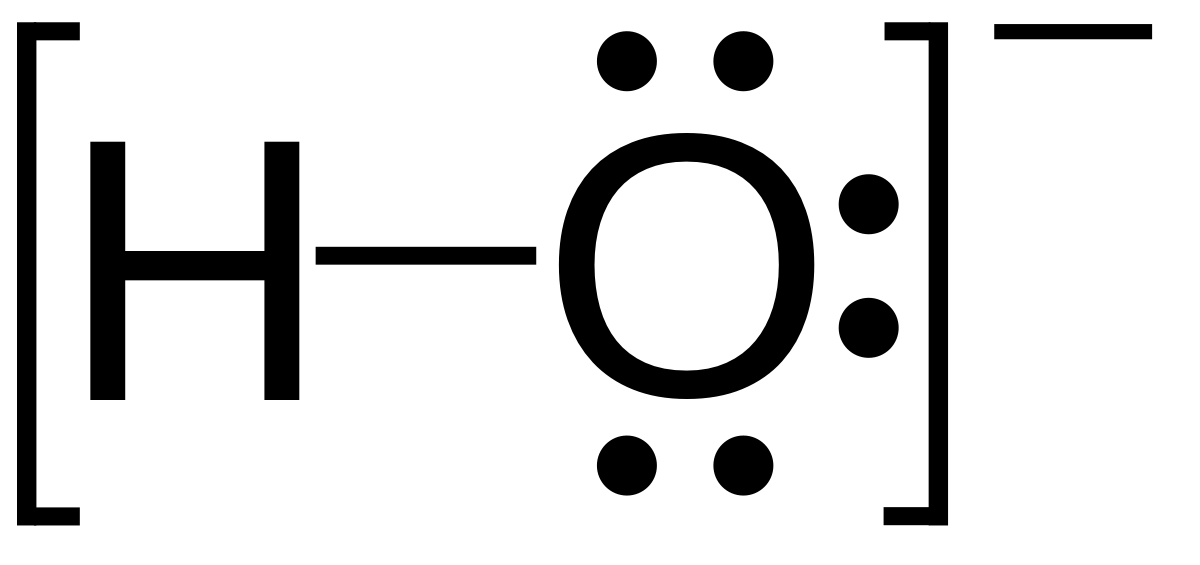
SOLVED: Determine the number of moles of oxygen atoms in each sample. a. 4.88 mol H2O2 b. 2.15 mol N2O c. 0.0237 mol H2CO3 d. 24.1 mol CO2

Moles”. “Moles” MOLE is a way of translating between grams and amu's a mole is a number that helps us translate between the atomic world and our. - ppt download

Energy level diagram for the molecular orbitals of OH ). H and O atom... | Download Scientific Diagram
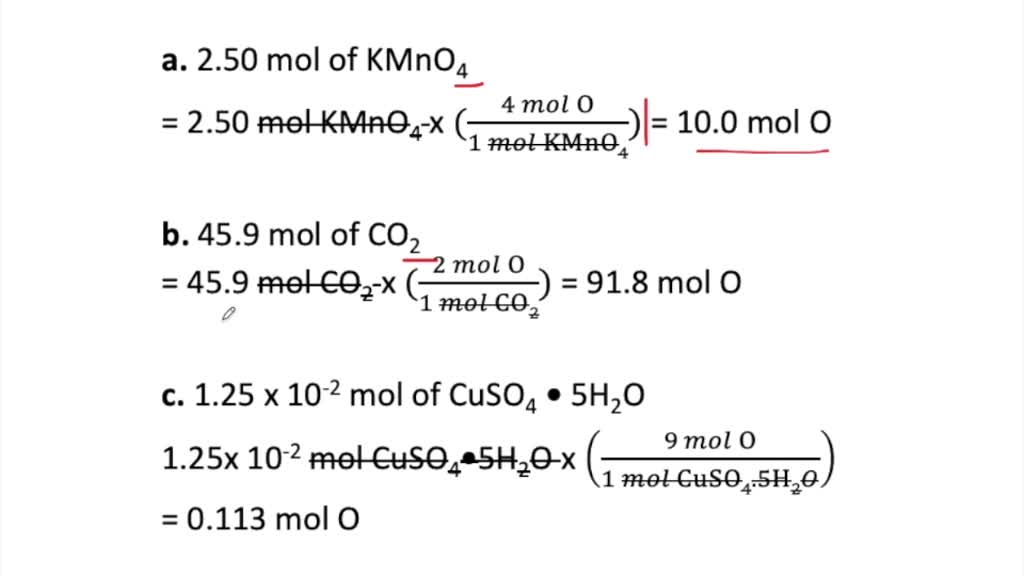
SOLVED:How many moles of oxygen atoms are contained in each compound? a. 2.50 mol of KMnO 4 b. 45.9 mol of CO2 c. 1.25 ×10^-2 mol of CuSO4 ·5 H2 O