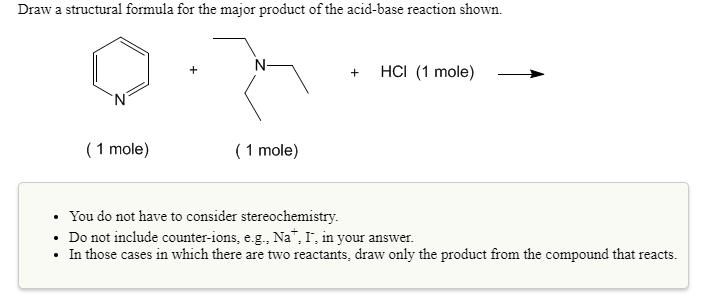
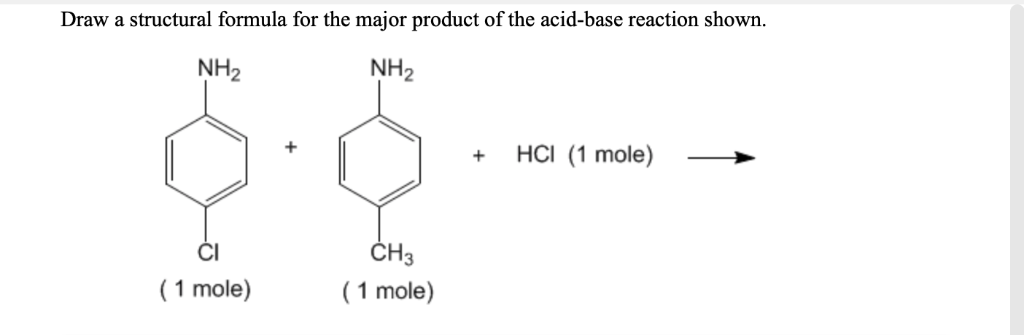
How many moles of magnesium chloride are formed when 55 mL of 0.70 M HCl is added to 42 mL of 1.204 g Mg(OH)_2? Mg(OH)_2(s) + 2HCl(aq) \rightarrow MgCl_2(aq) + 2H_2O(l) A.

Intro to Quantities Review Problems These are the calculations you should be able to perform: Sum of molar mass for a compound Convert mass mole Convert. - ppt download

SEM images of polished carbon steel surface before exposure to 1 mol.... | Download Scientific Diagram

What product will be obtained from the reaction of 1,3-cyclohexadiene with 1 mol HCl in ether? | Homework.Study.com